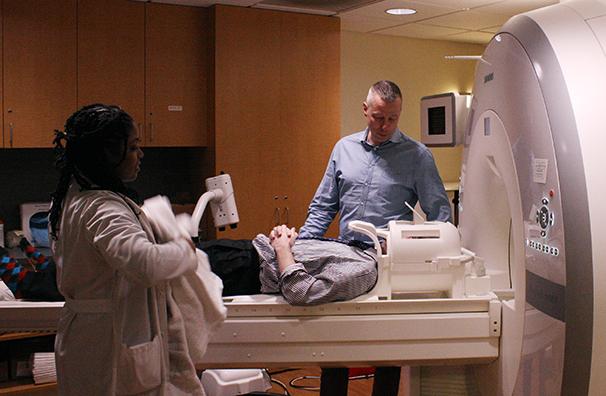
During the Canadian Depression Research and Intervention Network Conference on March 26 and 27, NYU psychiatry professor Alexander Neumeister announced the start of a research project aimed at finding a cure for Post-Traumatic Stress Disorder.
“The research was inspired by the fact that there is no evidence-based treatment developed specifically for PTSD patients,” Neumeister said.
Neumeister said the cause of PTSD is the lower level of cannabinoids, or neuro-chemicals that are found in the body as well as in marijuana.
Neumeister said there is a high incidence of people suffering from PTSD and using marijuana.
He concluded through a study done on animals that it might be more than the patients using marijuana in a recreational sense, but that they may be using it to replace the cannabinoids they are lacking.
“People with PTSD use marijuana at a very high rate,” Neumeister said. “It became very clear then, based on animal study, that it might be more than [being addicted to the] product because the animal studies showed that memory processes changed during exposure to marijuana.”
The research consisted of finding drugs that increase the levels of those neuro-chemicals.
“The new medications should help patients not necessarily forget but be less anxious, scared or threatened by the events they were exposed to,” Neumeister said.
Kate Hendry, a PTSD patient who attended the conference, said she was relieved by the current results of the research.
“I was happy to hear professor Neumeister state that current antidepressants don’t work very well for PTSD,” Hendry said. “I have been telling my doctors for years that medications don’t work for me, but that is always the first course of treatment.”
Brain-imaging studies have already been done on animals and humans to prove the relationship between cannabinoids and molecular changes. The next studies consist of verifying the efficacy of the drugs.
Two kinds of tests will be simultaneously conducted before the drug is released.
In June, one test will be done to be sure the drug is safe and tolerable, followed by clinical trials, analyses and reports to the U.S. Food and Drug Administration.
The other test will involve studying brain images to verify that the drug goes to human brains in sufficient concentrations and allows the increase in the amounts of cannabinoids needed for regular brain activity.
Hendry, who had several ineffective drugs prescribed to her in the past, said she is willing to overcome her distrust of health care and to help in clinical tests, hoping to find an effective treatment.
“I would be willing to participate in research or try the drug before its use becomes widespread, assuming I was also closely monitored and the risks of side effects were explained to me,” Hendry said.
CAS freshman Edward Mojica said the research for this drug could be life-saving for people who sacrifice their life for others.
“I think it’s a great use of research because so many veterans return home with that disorder,” Mojica said.
Neumeister said the drug should be available to PTSD patients within the next decade.
“We are still at the beginning of the process,” Neumeister said. “I would think in an ideal world, the drug will be available to everybody in about six to eight years from now.”
A version of this article appeared in the Thursday, April 3 print edition. Caren Doueiry is a contributing writer. Email her at [email protected]



































































































































Robert • May 10, 2014 at 11:38 am
Read, ‘PTSD/Bipolar Misdiagnosed’. It will explain what causes serotonin syndrome. It’s an infection. It can be tested.
Tom Cloyd • May 4, 2014 at 7:03 pm
I am appalled by so much in this article I hardly know where to start. We DO have well-validated, highly successful psychotherapy treatments for PTSD. We do NOT have medical treatments, and never will – it’s not a physical disorder. Why does this psychiatrist not know this?
There was a recent very major review of psychotherapy by a professional group in Australia. It is a superb source of first class information about what we know and can do with this disorder. I’ve written about this here:
http://thetraumapsych.wordpress.com/2013/09/24/update-ptsd-asd-treatment-guidelines-updated/
Diane • May 24, 2014 at 11:20 pm
Tom – you should review previous brain imaging studies. There is definitive evidence that there are physical changes to the brain directly related to PTSD.
There are a many well-documented cases of treatment resistant PTSD. This research is being conducted in order to address such cases.